You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
copper fermentation barrel
- Thread starter copperpot
- Start date

Help Support Winemaking Talk - Winemaking Forum:
This site may earn a commission from merchant affiliate
links, including eBay, Amazon, and others.
SouthernChemist
Professor
- Joined
- Nov 13, 2011
- Messages
- 738
- Reaction score
- 104
As you can tell I am new to this so here goes. Would it be OK to ferment in a copper drum?
This is just my chemistry instinct jumping out, I'm sure someone else can say for sure, but unless the drum has some sort of protective lining on it, it probably would not be safe fermenting an acidic mixture in a copper container. Even household vinegar will corrode copper over time, and that process will leach copper ions into your must.
Arne
Senior Member
I believe Southern Chemist is hittin the nail pretty close to on the head. I know you can use a crock for the primary, but if you want to move it around, a crock filled with 5 or6 gal of wine is pretty heavy. Food grade buckets are pretty cheap, and lots of people here have managed to get some free ones by going to the local bakery and getting their used frosting buckets. Clean em out good and they are ready to use. Arne.
I am not recommending the use of copper vats for fermentation. Now here comes the big BUT. I know they were used for fermenting "mash" for making illegal alcohol during Prohibition. However, I don't know whether or not mash was acidic or alkaline (basic), nor do I know if the distilliation process would eliminate any harmful substances from the mash.
pete1325
Senior Member
Is'nt distilling and fermenting two different process's?
Yes they are Pete. But in order to distill people that do/did that sort of thing first have to ferment a mash or corn, rye, wheat, etc. and then that fermented mash is distilled to produce alcohol. This, of course, is illegal in the United States but it is certainly part of our history, not to mention part of our present in some parts of the country.

$29.95 ($0.00 / Count)
$39.95 ($0.00 / Count)
Survival Vegetable Seeds Garden Kit Over 16,000 Seeds Non-GMO and Heirloom, Great for Emergency Bugout Survival Gear 35 Varieties Seeds for Planting Vegetables 35 Free Plant Markers Gardeners Basics
Home and Garden Meadows

$22.99
$40.00
The Sommelier's Atlas of Taste: A Field Guide to the Great Wines of Europe
Amazon.com

$175.50 ($175.50 / Count)
Wine Ingredient Kit - CRU SELECT Italian Style Sangiovese
Bridgeview Beer and Wine Supply

$170.00 ($170.00 / Count)
Wine Ingredient Kit - CRU SELECT Australia Style Chardonnay
Hobby Homebrew

$14.80
$24.00
The Geography of Wine: How Landscapes, Cultures, Terroir, and the Weather Make a Good Drop
Amazon.com

$29.99 ($15.00 / Count)
$41.80 ($20.90 / Count)
2 Pack 1 Gallon Large Fermentation Jars with 3 Airlocks and 2 SCREW Lids(100% Airtight Heavy Duty Lid w Silicone) - Wide Mouth Glass Jars w Scale Mark - Pickle Jars for Sauerkraut, Sourdough Starter
Qianfenie Direct

$129.00 ($129.00 / Count)
Cru International - Washington Merlot Style Wine Ingredient Kit
Hobby Homebrew

$175.50 ($175.50 / Count)
Wine Ingredient Kit - CRU SELECT Australia Style Viognier Pinot Gris
Bridgeview Beer and Wine Supply

$155.96 ($155.96 / Count)
Winexpert Revelation Napa Cabernet Sauvignon 3 Gallon (Limited) Wine Ingredient Kit
Discount Hydroponics LLc
Copper is poisonous in certain amounts in wine. There is an fda guideline for how much you can have. Copper sulfate is oftentimes used to reduce hydrogen sulfide in wine. The calculations for using copper sulfate specially takes this threshold into consideration when adding it to treat wine. You cannot gave more than X parts per million of copper. That being said you cannot estimate or measure how much copper would end up in your wine should you ferment in a copper container.
Short answer, no it would probably not be safe. Buy pails to ferment in or even in glass.
Short answer, no it would probably not be safe. Buy pails to ferment in or even in glass.
housecopper
researcher?
- Joined
- Jul 13, 2017
- Messages
- 15
- Reaction score
- 4
Hi guys - I'm in research mode on copper and wine making. Saw this thread and thought I'd make a comment...I don't believe there would be much copper leaching into the wine if you're not actually heating the wine. That's the whole point of lining copperware - if you're heating it, you need it lined (unless you're making jam/sugar/pastries or beating egg whites, etc), but otherwise copper's pretty much safe and doesn't really transfer any copper into food, for all the molecules may react with the contents. So the copper may 'react' with the wine, but it's not going to add copper to the wine unless you a) heat it or b) add copper sulfate (which is not the same thing as putting wine in a copper basin). Copper stills worked back in the day because of the process happening...AND you were heating the copper (I make vintage stills sometimes, so I get the mechanics) So again...you still need heat to make the inert copper crystals react with the food. Otherwise the copper walls are just "purifying" your wine - eg, cleaning out any bacteria, etc. Which you may or may not want. So perhaps using copper basins/kettles might work in terms of wine making...I'm not sure how or where in the process (I'm no wine maker, just a copper cookware fabricator) you'd use it, but it's something I'm certainly curious about in terms of the science... 
stickman
Veteran Winemaker
- Joined
- Jun 16, 2014
- Messages
- 1,930
- Reaction score
- 2,248
It really isn't a good idea to use copper for fermentation or storage. As others have already pointed out, copper will be released into the wine without control. Excess copper in the wine can cause several issues including premature oxidation, as well as haze or sediment in white wines.
Hi guys - I'm in research mode on copper and wine making. Saw this thread and thought I'd make a comment...I don't believe there would be much copper leaching into the wine if you're not actually heating the wine.
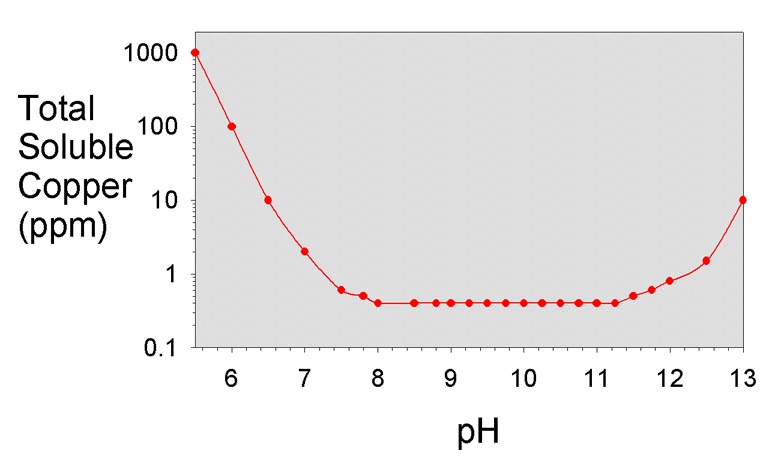
Housecopper, you do not seem to be aware of the effect of low pH on the solubility of copper. The dissolution rate of copper goes up dramatically at lower pH, and wine is rather acidic (pH ~ 3.2-3.5).
Last edited:
housecopper
researcher?
- Joined
- Jul 13, 2017
- Messages
- 15
- Reaction score
- 4
Hi stickman!
Thank you for the comments - as I mentioned, I'm just researching (not winemaking). I do know copper is not released into food unless the copper is heated (blame the day job as a copper cookware fabricator) and even then it's trace amounts - it takes a lot (years!) of cooking acidic foods in unlined copper for you to get an inkling of copper-induced illness. I do know liquids can be purified by sitting in copper, but it doesn't actually do much other than "clean" the liquid itself - hence ancient Egyptians would put water in copper jugs to purify it before drinking, etc. We get a lot of orders from people who want copper cups that they want water sitting in so they can drink naturally purified water...BUT that being said, I have no notion if something acidic, like wine, would react with unheated copper. It's an interesting experiment idea. I'm guessing you're right in that there would be some interaction, though I don't know if there'd be excess copper by any means...? Does anyone have numbers out there to show that wine sitting in copper would do anything? Perhaps eliminate the need for copper sulfate? And I don't know if it would be a good material for fermentation or storage - as I say, I know next to nothing about wine-making so far - but there was talk of using copper sheet in some other stages of wine making, so it got me thinking... Either way - thanks for humoring me on the research end...
Thank you for the comments - as I mentioned, I'm just researching (not winemaking). I do know copper is not released into food unless the copper is heated (blame the day job as a copper cookware fabricator) and even then it's trace amounts - it takes a lot (years!) of cooking acidic foods in unlined copper for you to get an inkling of copper-induced illness. I do know liquids can be purified by sitting in copper, but it doesn't actually do much other than "clean" the liquid itself - hence ancient Egyptians would put water in copper jugs to purify it before drinking, etc. We get a lot of orders from people who want copper cups that they want water sitting in so they can drink naturally purified water...BUT that being said, I have no notion if something acidic, like wine, would react with unheated copper. It's an interesting experiment idea. I'm guessing you're right in that there would be some interaction, though I don't know if there'd be excess copper by any means...? Does anyone have numbers out there to show that wine sitting in copper would do anything? Perhaps eliminate the need for copper sulfate? And I don't know if it would be a good material for fermentation or storage - as I say, I know next to nothing about wine-making so far - but there was talk of using copper sheet in some other stages of wine making, so it got me thinking... Either way - thanks for humoring me on the research end...
housecopper
researcher?
- Joined
- Jul 13, 2017
- Messages
- 15
- Reaction score
- 4
Housecopper, you do not seem to be aware of the effect of low pH on the solubility of copper. The dissolution rate of copper goes up dramatically at lower pH, and wine is rather acidic (pH ~ 3.2-3.5).
Nope, totally not aware of the low pH interaction with copper. I know all about how copper interacts with food and liquids when the copper is heated, but not when it simply is in contact with acid. I definitely will research!! Thank you!
What about the whole running wine over copper sheet to get rid of the sulfur smell? I swear I read that somewhere...here? So...there's some merit to using copper sheet somewhere in the process? Just very fascinating.
housecopper
researcher?
- Joined
- Jul 13, 2017
- Messages
- 15
- Reaction score
- 4
I think I'll go read a lot of copper plumbing manuals to get some additional ideas too and learn about cold/non-heat induced solubility. Still have some notions where copper and wine can play together, though. Could be cool/interesting. Why not, right?
Scooter68
Fruit "Wine" Maker
Let's not neglect to mention that in addition to the acidic nature of a wine must you also have the chemicals added Potassium Metabisulfate, and Potasium Sorbate. Even if the copper doesn't contribute to a toxic condition it may affect the ability of those chemicals to do their job. (Isn't whipping a copper wire in a wine must a recommended treatment for too much HS2?)
Over the centuries of wine making copper has been around and while not cheap, one would expect there is a reason it's not mentioned as good material for wine fermentation.
Over the centuries of wine making copper has been around and while not cheap, one would expect there is a reason it's not mentioned as good material for wine fermentation.
Copper compounds are TOXIC . It is one thing to use copper pipes to carry chemically neutral water, and another thing to expose fermenting wine with all complex acids etc etc. to a copper walls of a barrel.
I do not want any copper touching my wine, period.
Beside copper is expensive comparing o other and SAFER alternatives like stainless steel, glass, or wood.
So why bother with copper?
I do not want any copper touching my wine, period.
Beside copper is expensive comparing o other and SAFER alternatives like stainless steel, glass, or wood.
So why bother with copper?
What about the whole running wine over copper sheet to get rid of the sulfur smell? I swear I read that somewhere...here? So...there's some merit to using copper sheet somewhere in the process? Just very fascinating.
Yes, H2S (and also more complex thiols AKA mercaptans) bind strongly to copper, so you can squelch the stinky smell by exposing the wine to copper. However, there will be a concomitant increase in copper dissolved into the wine. At low concentrations, this is fine. However, the problem is that the copper dose will be uncontrolled.
Instead, it is better in these situations to use Reduless, which introduces a known, safe quantity of copper.
In general, I am a huge fan of copper, and am not fearful of it in almost all circumstances. However, the solubility of copper at low pH makes me nervous.
Scooter68
Fruit "Wine" Maker
So for the safety of all who might consider it I believe it's the safe assumption to say Copper as a fermentation container for wine is way too risky even if it is coated since coatings can deteriorate and suddenly bring wine into contact with copper leading to unknown results and potentially even deadly results.
Perhaps this was tried before a hundred or more years past and the person doing the experimenting did not survive to report their failure. Would not be the first time people unknowingly did themselves in. Sort of like the folks in Hawaii trimming the leaves off Oleander branches and using them to roast hotdogs. Most did not live to say "Hmm bad idea."
As the Great Investigator Clouseau would say the case is Solv-ed.
Perhaps this was tried before a hundred or more years past and the person doing the experimenting did not survive to report their failure. Would not be the first time people unknowingly did themselves in. Sort of like the folks in Hawaii trimming the leaves off Oleander branches and using them to roast hotdogs. Most did not live to say "Hmm bad idea."
As the Great Investigator Clouseau would say the case is Solv-ed.
SouthernChemist
Professor
- Joined
- Nov 13, 2011
- Messages
- 738
- Reaction score
- 104
I would think it's simply not worth the risk. There are plenty of other containers you can ferment in that are safer.
Let's not forget that the Romans made wine in lead vessels. They used to boil grape must down in lead containers to concentrate it and inevitably let the lead affect the flavor. They also boiled old wine with other ingredients in lead vessels to improve its flavor. Oh the good ole days when people did not know any better!
Let's not forget that the Romans made wine in lead vessels. They used to boil grape must down in lead containers to concentrate it and inevitably let the lead affect the flavor. They also boiled old wine with other ingredients in lead vessels to improve its flavor. Oh the good ole days when people did not know any better!
Scooter68
Fruit "Wine" Maker
I would think it's simply not worth the risk. There are plenty of other containers you can ferment in that are safer.
Let's not forget that the Romans made wine in lead vessels. They used to boil grape must down in lead containers to concentrate it and inevitably let the lead affect the flavor. They also boiled old wine with other ingredients in lead vessels to improve its flavor. Oh the good ole days when people did not know any better!
So that means there was a reason the Roman leaders became complete idiots - Brain-damaged by the "Best Wine" the strongest flavor carrying a good dose of lead which can result in:
High blood pressure
Joint and muscle pain
Difficulties with memory or concentration
Headache
Abdominal pain
Mood disorders
Reduced sperm count and abnormal sperm
Miscarriage, stillbirth or premature birth in pregnant women
Similar threads
- Replies
- 9
- Views
- 660
- Replies
- 9
- Views
- 595
- Replies
- 18
- Views
- 915




































![[Upgraded] 9Pcs Tree Root Growing Box with Drain Holes, Half Transparent Plant Rooting Propagation Ball & Metal Core Twist Ties, for Fast Propagation Plants (Size M)](https://m.media-amazon.com/images/I/514MWQxtWOL._SL500_.jpg)






